1 Friedman DS, O’Colmain BJ, Munoz B, Tomany SC, McCarty C, De Jong PT, Nemesure B, Mitchell P, Kempen J. Prevalence of age-related macular degeneration in the United States. Arch ophthalmol. 2004 Apr 1;122(4):564-72.
2 Sobrin L, Seddon JM. Nature and nurture-genes and environment-predict onset and progression of macular degeneration. Progress in retinal and eye research. 2014 May 1;40:1-5.
3 Rosenfield PJ, Martidis A, Tennant M. Age-related macular degeneration. In: Yanoff M, Duker JS, Augsburger JJ, eds. Ophthalmology: Expert Consult. 3rd ed. Philadelphia, PA: Elsevier Mosby; 2009.
4 Shalev V, Sror M, Goldshtein I, Kokia E, Chodick G. Statin use and the risk of age related macular degeneration in a large health organization in Israel. Ophthalmic epidemiology. 2011 Apr 1;18(2):83-90.
5 Wong WL, Su X, Li X, Cheung CM, Klein R, Cheng CY, Wong TY. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. The Lancet Global Health. 2014 Feb 1;2(2):e106-16.
6 Ferris FL, Fine SL, Hyman L. Age-related macular degeneration and blindness due to neovascular maculopathy. Archives of ophthalmology. 1984 Nov 1;102(11):1640-2.
7 Cheung LK, Eaton A. Age‐related macular degeneration. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy. 2013 Aug;33(8):838-55.
8 Delcourt C, Souied E, Sanchez A, Bandello F. Development and validation of a risk score for age-related macular degeneration: the STARS Questionnaire. Investigative Ophthalmology & Visual Science. 2017 Dec 1;58(14):6399-407.
9 Bucan K, Lukic M, Bosnar D, Kopic A, Jukic T, Konjevoda S, Glavadanovic S, Gverovic Antunica A. Analysis of association of risk factors for age-related macular degeneration. European Journal of Ophthalmology. 2022 Jan;32(1):410-6.
10 Klein RJ, Zeiss C, Chew EY, Tsai JY, Sackler RS, Haynes C, Henning AK, SanGiovanni JP, Mane SM, Mayne ST, Bracken MB. Complement factor H polymorphism in age-related macular degeneration. Science. 2005 Apr 15;308(5720):385-9.
11 Bressler NM, Bressler SB, Fine SL. Age-related macular degeneration. Survey of ophthalmology. 1988 May 1;32(6):375-413.
12 Thomas MA, Grand MG, Williams DF, Lee CM, Pesin SR, Lowe MA. Surgical management of subfoveal choroidal neovascularization. Ophthalmology. 1992 Jun 1;99(6):952-68.
13 Ciardella AP, Donsoff IM, Huang SJ, Costa DL, Yannuzzi LA. Polypoidal choroidal vasculopathy. Survey of ophthalmology. 2004 Jan 1;49(1):25-37.
14 Imamura Y, Engelbert M, Iida T, Freund KB, Yannuzzi LA. Polypoidal choroidal vasculopathy: a review. Survey of ophthalmology. 2010 Nov 1;55(6):501-15.
15 Yannuzzi LA, Ciardella A, Spaide RF, Rabb M, Freund KB, Orlock DA. The expanding clinical spectrum of idiopathic polypoidal choroidal vasculopathy. Archives of ophthalmology. 1997 Apr 1;115(4):478-85.
16 Fong DS. Age-related macular degeneration: update for primary care. American Family Physician. 2000 May 15;61(10):3035-42.
17 El Baba F, Jarrett II WH, Harbin Jr TS, Fine SL, Michels RG, Schachat AP, Green WR. Massive hemorrhage complicating age-related macular degeneration: clinicopathologic correlation and role of anticoagulants. Ophthalmology. 1986 Dec 1;93(12):1581-92.
18 Yeo JH, Marcus S, Murphy RP. Retinal pigment epithelial tears: patterns and prognosis. Ophthalmology. 1988 Jan 1;95(1):8-13.
19 GREEN SN, YARIAN D. Acute tear of the retinal pigment epithelium. Retina. 1983 Jan 1;3(1):16-20.
20 https://eyewiki.aao.org/Age-Related_Macular_Degeneration#cite_note-53
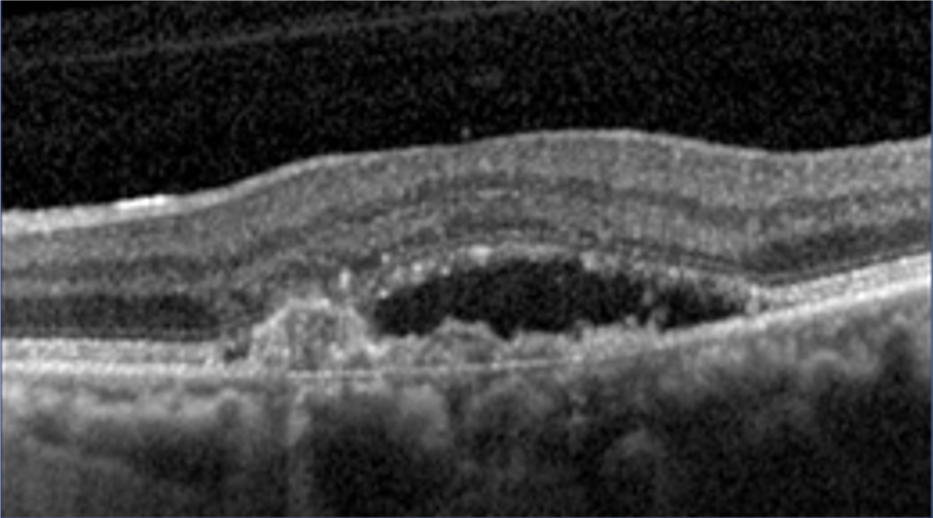
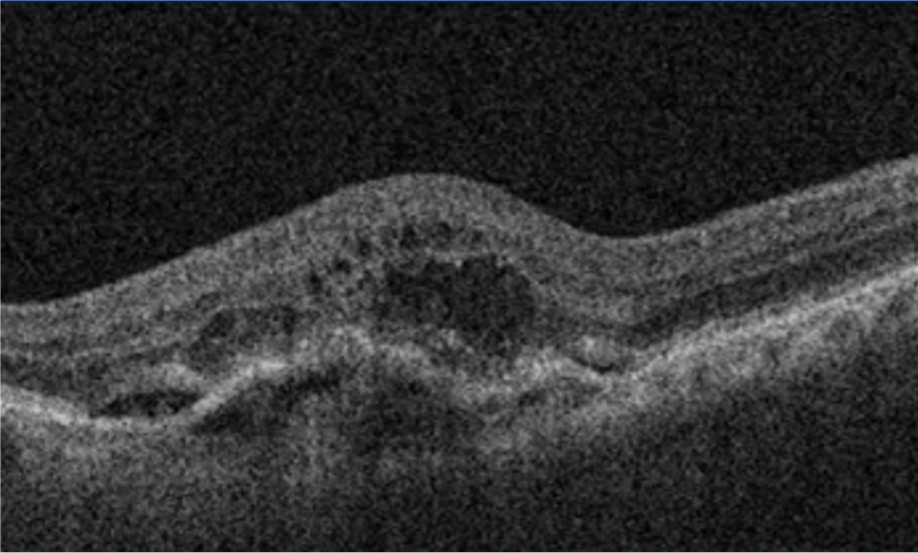
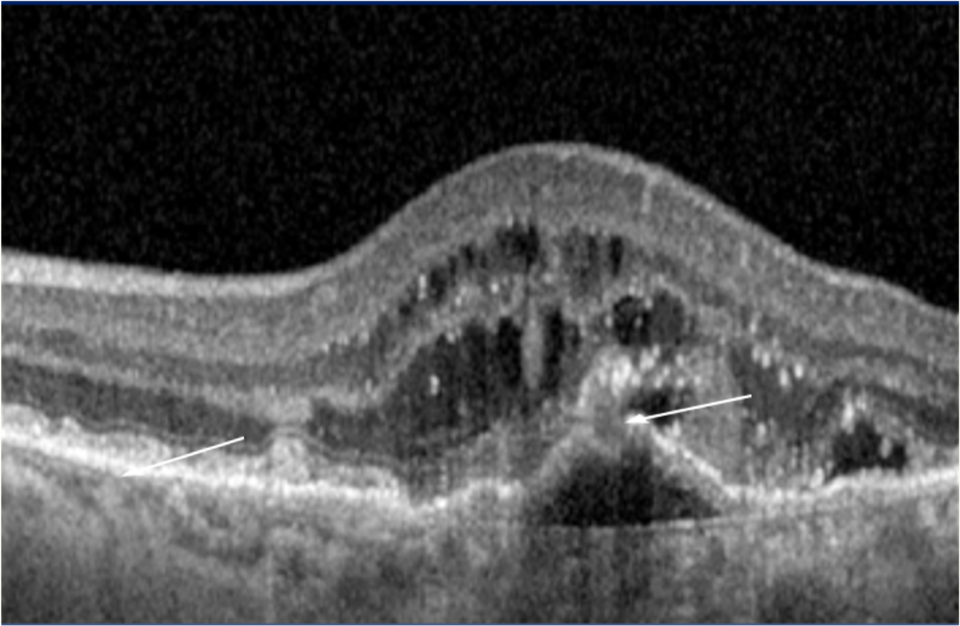
21 Alshahrani ST, Al Shamsi HN, Kahtani ES, Ghazi NG. Spectral-domain optical coherence tomography findings in polypoidal choroidal vasculopathy suggest a type 1 neovascular growth pattern. Clinical Ophthalmology (Auckland, NZ). 2014;8:1689.
22 Koustenis A, Harris A, Gross J, Januleviciene I, Shah A, Siesky B. Optical coherence tomography angiography: an overview of the technology and an assessment of applications for clinical research. British Journal of Ophthalmology. 2017 Jan 1;101(1):16-20.
23 Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, Kim RY. Ranibizumab for neovascular age-related macular degeneration. New England Journal of Medicine. 2006 Oct 5;355(14):1419-31.
24 Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, Kim RY. Ranibizumab for neovascular age-related macular degeneration. New England Journal of Medicine. 2006 Oct 5;355(14):1419-31.
25 Chhablani J, Paulose RM, Lasave AF, Wu L, Carpentier C, Maia M, Lujan S, Rojas S, Serrano M, Berrocal MH, Arevalo JF. Intravitreal bevacizumab monotherapy in myopic choroidal neovascularisation: 5-year outcomes for the PAN-American Collaborative Retina Study Group. British Journal of Ophthalmology. 2018 Apr 1;102(4):455-9.
26 Brown DM, Tuomi L, Shapiro H, Pier Study Group. Anatomical measures as predictors of visual outcomes in ranibizumab-treated eyes with neovascular age-related macular degeneration. Retina. 2013 Jan 1;33(1):23-34.
27 Heier JS, Brown DM, Chong V, Korobelnik JF, Kaiser PK, Nguyen QD, Kirchhof B, Ho A, Ogura Y, Yancopoulos GD, Stahl N. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology. 2012 Dec 1;119(12):2537-48.
28 Schmidt-Erfurth U, Kaiser PK, Korobelnik JF, Brown DM, Chong V, Nguyen QD, Ho AC, Ogura Y, Simader C, Jaffe GJ, Slakter JS. Intravitreal aflibercept injection for neovascular age-related macular degeneration: ninety-six–week results of the VIEW studies. Ophthalmology. 2014 Jan 1;121(1):193-201.
29 Singer MA, Awh CC, Sadda S, Freeman WR, Antoszyk AN, Wong P, Tuomi L. HORIZON: an open-label extension trial of ranibizumab for choroidal neovascularization secondary to age-related macular degeneration. Ophthalmology. 2012 Jun 1;119(6):1175-83.
30 Rosenfeld PJ, Shapiro H, Tuomi L, Webster M, Elledge J, Blodi B. Characteristics of patients losing vision after 2 years of monthly dosing in the phase III ranibizumab clinical trials. Ophthalmology. 2011 Mar 1;118(3):523-30.
31 Wolf S, Holz FG, Korobelnik JF, Lanzetta P, Mitchell P, Prünte C, Schmidt-Erfurth U, Weichselberger A, Hashad Y. Outcomes following three-line vision loss during treatment of neovascular age-related macular degeneration: subgroup analyses from MARINA and ANCHOR. British journal of ophthalmology. 2011 Dec 1;95(12):1713-8.
32 Tadayoni R, Sararols L, Weissgerber G, Verma R, Clemens A, Holz FG. Brolucizumab: a newly developed anti-VEGF molecule for the treatment of neovascular age-related macular degeneration. Ophthalmologica. 2021;244(2):93-101.
33 Khanani AM, Patel SS, Ferrone PJ, Osborne A, Sahni J, Grzeschik S, Basu K, Ehrlich JS, Haskova Z, Dugel PU. Efficacy of every four monthly and quarterly dosing of faricimab vs ranibizumab in neovascular age-related macular degeneration: the STAIRWAY phase 2 randomized clinical trial. JAMA ophthalmology. 2020 Sep 1;138(9):964-72.
34 Lukic M, Eleftheriadou M, Hamilton RD, Rajendram R, Bucan K, Patel PJ. Four-year outcomes of aflibercept treatment for neovascular age-related macular degeneration: Results from real-life setting. European Journal of Ophthalmology. 2021 Jul;31(4):1940-4.
35 Eleftheriadou M, Gemenetzi M, Lukic M, Sivaprasad S, Hykin PG, Hamilton RD, Rajendram R, Tufail A, Patel PJ. Three-year outcomes of aflibercept treatment for neovascular age-related macular degeneration: evidence from a clinical setting. Ophthalmology and therapy. 2018 Dec;7(2):361-8.
36 Augsburger M, Sarra GM, Imesch P. Treat and extend versus pro re nata regimens of ranibizumab and aflibercept in neovascular age-related macular degeneration: a comparative study. Graefes Arch Clin Exp Ophthalmol. 2019 Sep;257(9):1889-1895. doi: 10.1007/s00417-019-04404-0. Epub 2019 Jun 29. PMID: 31256237.