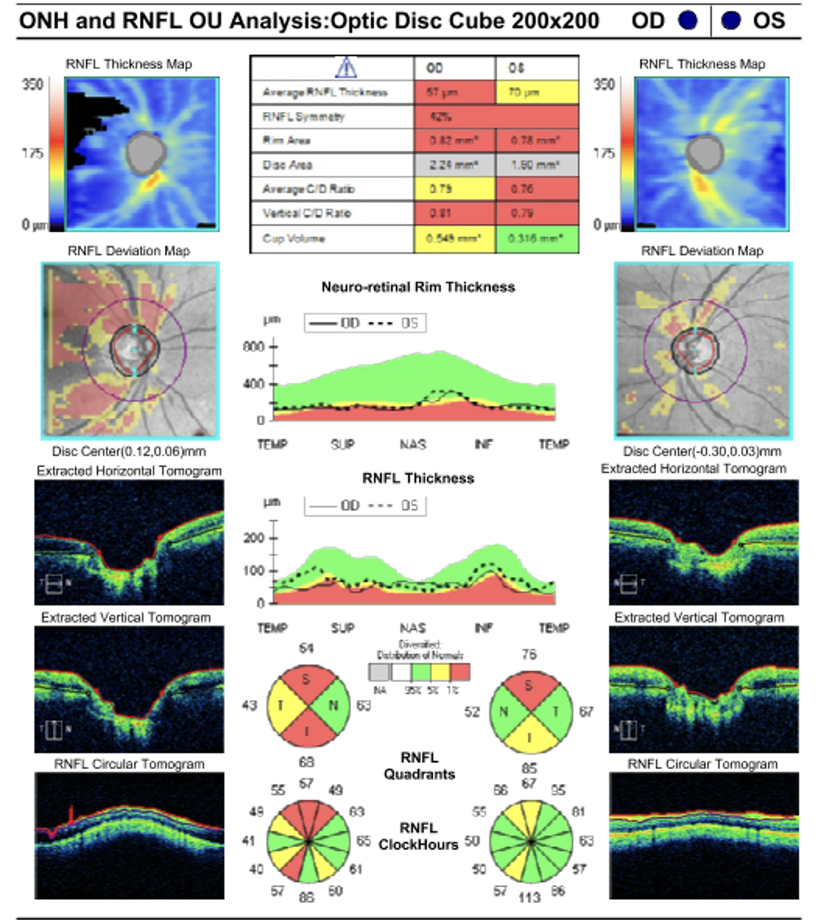
Glaucoma is a big public health problem. It is the leading cause of irreversible blindness worldwide and is associated with a reduced quality of life.(1) An estimated 57.5 million people worldwide are affected by primary open-angle Glaucoma (POAG), with a global prevalence of 2.2%.(2) In Europe, 7.8 million people were affected by POAG, and the prevalence is 2.51%.(3-5)
Glaucoma describes a group of progressive conditions with characteristic cupping of the optic disc with corresponding visual field defects due to retinal ganglion cell loss.(6) Primary open-angle glaucoma (POAG) is a subset, defined by an open, normal-appearing anterior chamber angle and raised intraocular pressure (IOP) with no other underlying disease.(6,7) If there is an identifiable underlying cause for raised IOP, this is termed secondary open-angle glaucoma. If the IOP is within normal limits, this is termed normal-tension glaucoma (NTG).(6,7)
Several risk factors are identified for POAG: IOP, age, race, corneal thickness, and family history. The most important one is intraocular pressure, which is also the only that can be modified, compared to the other risk factors. Patients with ocular hypertension, who have an increased intraocular pressure but no damage to optic discs/visual fields, have a higher risk of developing glaucoma, as per the Ocular Hypertension Treatment Study and European Glaucoma Prevention Study.(8,9) Furthermore, both studies revealed that the incidence of POAG increases with age. (8,9) Several studies have shown POAG to be more prevalent in people of African-Caribbean descent than Caucasians. In addition, ocular hypertension is more likely to progress in POAG in patients of African-Caribbean descent.(10,11) If some has a first-degree relative with POAG, they have a higher risk for developing POAG.(12,13) This has been reported in several studies, with the odds ratio varying from 3 to 13.(7) There is controversy and no consensus on whether myopia, hypertension, and diabetes are risk factors.
There are multiple mechanisms that explain the pathomechanism of glaucoma development. Some are IOP-dependent, and some are non-IOP-dependent. There is consensus that increased IOP is secondary to obstruction of trabecular meshwork flow rather than hyperproduction of the aqueous humour. The elevated pressure either causes mechanical damage to the optic nerve head at the lamina cribrosa or causes vascular damage, which causes ischaemia. The obstructed trabecular meshwork may be a result of several mechanisms:
- trabecular meshwork obstruction with foreign material
- trabecular endothelial cell loss
- loss of trabecular endothelial cell phagocytic activity
- loss of giant vacuoles of the endothelium of Schlemm’s canal
- loss of pore density and reduced pore size in the wall of the Schlemm’s canal
The non-IOP-dependent mechanisms are:
- reduced ocular perfusion pressure
- excitotoxic damage from excessive glutamate
- autoimmune damage
- loss of protective neurotrophic factors
- failure of cellular repair mechanisms
- abnormal regulation of choroidal and retinal vasculature
Normal-tension glaucoma (NTG)
Numerous studies revealed and calculated the physiologic mean of the IOP.(14-18) As per consensus, the upper limit of the normal IOP is 21 mmHg. However, the Baltimore Eye Survey identified that 50% of the patients with POAG had an IOP lower than 22 mmHg.(19) Several mechanisms (non-IOP related) have been proposed as important in the pathophysiology of NTG: systemic and local vascular dysregulation, hematologic abnormalities, impaired cerebrospinal fluid (CSF) circulation resulting in stagnation and decreased optic nerve protection, and structural anomalies, including structural weakness of the lamina cribrosa. Nonetheless, the Collaborative Normal Tension Study showed a slowing of the progression of the disease in patients who had achieved a 30% or more reduction of an already normal IOP.(20,21)
Secondary open-angle glaucoma (SOAG)
Secondary open-angle glaucoma is any form of glaucoma with an identifiable cause of increased eye pressure, resulting in optic nerve damage and vision loss. It may be caused by an eye injury, inflammation, certain drugs such as steroids, and advanced cases of cataracts or diabetes.(22) Below is a list of types that represent secondary open-angle glaucomas:
- Pseudo-exfoliative glaucoma is present in patients with the pseudo-exfoliative syndrome. It is a systemic condition characterised by the deposition of white dandruff-like material, called exfoliation material, within the anterior segment of the eye and other organs such as the heart, lungs, and kidneys. 50% of patients with exfoliation syndrome are ultimately diagnosed with glaucoma. It is often unilateral and slowly progressive; therefore, patients present at a late stage of the disease.(23)
- Neovascular glaucoma (open-angle stage) occurs in patients with severe retinal ischaemia (secondary to proliferative diabetic retinopathy, ischaemic central retinal vein occlusion, and ischaemic ocular syndrome). Read more in this chapter.
- Steroid-induced glaucoma – The use of topical and systemic corticosteroids for a longer time can result in a rise in intraocular pressure, which is usually reversible once the patient stops using the medication.(24)
- Pigmentary glaucoma – In certain eyes, pigment particles may circulate abnormally in the aqueous fluid, which may cause blockage at the drainage angle.(24)
- Traumatic glaucoma is caused by trauma. One of the mechanisms is the blockage of trabecular meshwork by blood.
- Lens-induced phacolyitic glaucoma – Lens material may cause blockage of the outflow of the aqueous at the drainage angle, which may occur after injury (including cataract surgery) or when lens material leaks through the lens capsule of a mature/hypermature lens.(24)
- Uveitic glaucoma – In uveitis, cells and proteins in the anterior chamber disturb the normal outflow of aqueous fluid through the trabecular meshwork, causing a raised intraocular pressure.(24)
- Congenital glaucoma is developmental glaucoma that occurs before the age of three due to an obstruction that prevents adequate drainage of aqueous humour, caused by abnormal development of the trabecular meshwork and the anterior chamber angle.(25)